Abstract
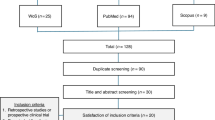
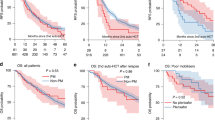
The use of non-cryopreserved hematopoietic stem cells (HSC) can be an alternative to the traditional cryopreserved infusions of HSCs in autologous stem cell transplantation (aHSCT). After high-dose melphalan conditioning (HDM), we sought to compare time to engraftment, overall survival, and safety in multiple myeloma (MM) patients undergoing a first aHSCT after high-dose melphalan conditioning (HDM). We conducted a cohort study from March 2018 to December 2019. Of all autologous transplants performed during this period, 105 were for MM as the first consolidation. Fifty-one patients received a cryopreserved graft; the remaining 54 patients received a fresh infusion. General clinical characteristics were similar between these two groups. Cell viability was higher in non-cryopreserved grafts (95% vs. 86% p < 0.01). Four deaths occurred during hospitalization in the cryopreserved group, one in the non-cryopreserved group. The cumulative incidence of neutrophil and platelet engraftment on D + 25 was higher in the non-cryopreserved compared to the cryopreserved group (98% vs 90% p < 0.01 and 96.2% vs 72.54% p < 0.01 respectively). Additionally, the hospital length of stay was reduced by 4 days for patients for the non-cryopreserved cohort. In summary, the use of non-cryopreserved HSCs after HDM is safe and effective compared to patients who received a cryopreserved graft.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Our dataset is available and will be provided under requested supported by adequate justification.
References
Horowitz M, Schreiber H, Elder A, Heidenreich O, Vormoor J, Toffalori C, et al. Epidemiology and Biology of Relapse after Stem Cell Transplantation. Bone Marrow Transpl. 2018;53:1379–89.
Dhakal B, Szabo A, Chhabra S, Hamadani M, D’Souza A, Usmani SZ, et al. Autologous Transplantation for Newly Diagnosed Multiple Myeloma in the Era of Novel Agent Induction: A Systematic Review and Meta-analysis. JAMA Oncol. 2018;4:343–50.
Attal M, Harousseau J-L, Stoppa A-M, Sotto J-J, Fuzibet J-G, Rossi J-F, et al. A Prospective, Randomized Trial of Autologous Bone Marrow Transplantation and Chemotherapy in Multiple Myeloma. N. Engl J Med. 1996;335:91–7.
Kayal S, Sharma A, Iqbal S, Tejomurtula T, Cyriac SL, Raina V. High-dose chemotherapy and autologous stem cell transplantation in multiple myeloma: a single institution experience at All India Institute of Medical Sciences, New Delhi, using non-cryopreserved peripheral blood stem cells. Clin Lymphoma Myeloma Leuk. 2014;14:140–7.
Sarmiento M, Ramírez P, Parody R, Salas MQ, Beffermann N, Jara V, et al. Advantages of non-cryopreserved autologous hematopoietic stem cell transplantation against a cryopreserved strategy. Bone Marrow Transpl. 2018;53:960–6.
Bittencourt MCB, Mariano L, Moreira F, Schmidt-Filho J, Mendrone-Jr A, Rocha V. Cryopreserved versus non-cryopreserved peripheral blood stem cells for autologous transplantation after high-dose Melphalan in multiple myeloma: comparative analysis. Bone Marrow Transpl. 2019;54:138–41.
Ruiz-Argüelles GJ, Steensma DP. Staunching the rising costs of haematological health care. Lancet Haematol. 2016;3:e455.
Kardduss-Urueta A, Gale RP, Gutierrez-Aguirre CH, Herrera-Rojas MA, Murrieta-Álvarez I, Perez-Fontalvo R, et al. Freezing the graft is not necessary for autotransplants for plasma cell myeloma and lymphomas. Bone Marrow Transpl. 2018;53:457–60.
Wannesson L, Panzarella T, Mikhael J, Keating A. Feasibility and safety of autotransplants with noncryopreserved marrow or peripheral blood stem cells: a systematic review. Ann Oncol J Eur Soc Med Oncol. 2007;18:623–32.
Ager S, Scott MA, Mahendra P, Richards EM, Jestice HK, Boraks P, et al. Peripheral blood stem cell transplantation after high-dose therapy in patients with malignant lymphoma: a retrospective comparison with autologous bone marrow transplantation. Bone Marrow Transpl. 1995;16:79–83.
Cuellar-Ambrosi F, Karduss UA, Gomez WR, Mondragon MC, Velasquez-Lopera M, Calle S. Hematologic reconstitution following high-dose and supralethal chemoradiotherapy using stored, noncryopreserved autologous hematopoietic stem cells. Transpl Proc. 2004;36:1704–5.
Ruiz-Argüelles GJ, León-Peña AA, León-González M, Nuñez-Cortes AK, Olivares-Gazca JC, Murrieta-Alvarez I, et al. A Feasibility Study of the Full Outpatient Conduction of Hematopoietic Transplants in Persons with Multiple Sclerosis Employing Autologous Non-Cryopreserved Peripheral Blood Stem Cells. Acta Haematol. 2017;137:214–9.
Ruiz-Argüelles GJ, Gómez-Rangel D, Ruiz-Delgado GJ, Ruiz-Argüelles A, Pérez-Romano B, Rivadeneyra L. Results of an autologous noncryopreserved, unmanipulated peripheral blood hematopoietic stem cell transplant program: a single-institution, 10-year experience. Acta Haematol. 2003;110:179–83.
Kingston JE, Malpas JS, Stiller CA, Pritchard J, McElwain TJ. Autologous bone marrow transplantation contributes to haemopoietic recovery in children with solid tumours treated with high dose melphalan. Br J Haematol. 1984;58:589–95.
Carella AM, Santini G, Santoro A, Coser P, Frassoni F, Martinengo M, et al. Massive chemotherapy with non-frozen autologous bone marrow transplantation in 13 cases of refractory Hodgkin’s disease. Eur J Cancer Clin Oncol. 1985;21:607–13.
Russell JA, Selby PJ, Ruether BA, Mbidde EK, Ashley S, Zulian G, et al. Treatment of advanced Hodgkin’s disease with high dose melphalan and autologous bone marrow transplantation. Bone Marrow Transpl. 1989;4:425–9.
Carey PJ, Proctor SJ, Taylor P, Hamilton PJ. Autologous bone marrow transplantation for high-grade lymphoid malignancy using melphalan/irradiation conditioning without marrow purging or cryopreservation. The Northern Regional Bone Marrow Transplant Group. Blood 1991;77:1593–8.
Sierra J, Conde E, Iriondo A, Brunet S, Marín J, Pérez de Oteiza J, et al. Frozen vs. nonfrozen bone marrow for autologous transplantation in lymphomas: a report from the Spanish GEL/TAMO Cooperative Group. Ann Hematol. 1993;67:111–4.
Taylor PR, Jackson GH, Lennard AL, Lucraft H, Proctor SJ. Autologous transplantation in poor risk Hodgkin’s disease using high dose melphalan/etoposide conditioning with non-cryopreserved marrow rescue. The Newcastle and Northern Region Lymphoma Group. Br J Cancer. 1993;67:383–7.
Holowiecki J, Giebel S, Wojnar J, Krawczyk-Kulis M, Stella-Holowiecka B, Kachel L, et al. Autologous hematopoietic stem cell transplantation for high-risk Hodgkin’s disease: a single-center experience with the first 100 patients. Transpl Proc. 2002;34:3378–83.
Jones N, Williams D, Broadbent V, Jestice K, Boraks P, Scott M, et al. High-dose melphalan followed by autograft employing non-cryopreserved peripheral blood progenitor cells in children. Eur J Cancer Oxf Engl 1990. 1996;32A:1938–42.
Papadimitriou CA, Dimopoulos MA, Kouvelis V, Kostis E, Kapsimali V, Contoyannis D, et al. Non-cryopreserved peripheral blood progenitor cells collected by a single very large-volume leukapheresis: a simplified and effective procedure for support of high-dose chemotherapy. J Clin Apheresis. 2000;15:236–41.
Mabed M, Al-Kgodary T. Cyclophosphamide, etoposide and carboplatine plus non-cryopreserved autologous peripheral blood stem cell transplantation rescue for patients with refractory or relapsed non-Hodgkin’s lymphomas. Bone Marrow Transpl. 2006;37:739–43.
Berro M, Chhabra S, Piñana JL, Arbelbide J, Rivas MM, Basquiera AL, et al. Predicting Mortality after Autologous Transplant: Development of a Novel Risk Score. Biol Blood Marrow Transpl. 2020;26:1828–32.
McCarthy PL, Hahn T, Hassebroek A, Bredeson C, Gajewski J, Hale G, et al. Trends in use of and survival after autologous hematopoietic cell transplantation in North America, 1995-2005: significant improvement in survival for lymphoma and myeloma during a period of increasing recipient age. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2013;19:1116–23.
Preussler JM, Denzen EM, Majhail NS. Costs and cost-effectiveness of hematopoietic cell transplantation. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2012;18:1620–8.
Tai B-C, Grundy RG, Machin D. On the importance of accounting for competing risks in pediatric cancer trials designed to delay or avoid radiotherapy: I. Basic concepts and first analyses. Int J Radiat Oncol Biol Phys. 2010;76:1493–9.
Majolino I, Mohammed D, Hassan D, Ipsevich F, Abdullah C, Mohammed R, et al. Initial Results of Peripheral-Blood Stem-Cell Mobilization, Collection, Cryopreservation, and Engraftment After Autologous Transplantation Confirm That the Capacity-Building Approach Offers Good Chances of Success in Critical Contexts: A Kurdish-Italian Cooperative Project at the Hiwa Cancer Hospital, Sulaymaniyah. J Glob Oncol. 2018;4:1–8.
Blommestein HM, Verelst SGR, Huijgens PC, Blijlevens NMA, Cornelissen JJ, Uyl-de Groot CA. Real-world costs of autologous and allogeneic stem cell transplantations for haematological diseases: a multicentre study. Ann Hematol. 2012;91:1945–52.
Mishra V, Andresen S, Brinch L, Kvaløy S, Ernst P, Lønset MK, et al. Cost of autologous peripheral blood stem cell transplantation: the Norwegian experience from a multicenter cost study. Bone Marrow Transpl. 2005;35:1149–53.
Faucher C, Le Corroller Soriano AG, Esterni B, Vey N, Stoppa AM, Chabannon C, et al. Randomized study of early hospital discharge following autologous blood SCT: medical outcomes and hospital costs. Bone Marrow Transpl. 2012;47:549–55.
van Agthoven M, Vellenga E, Fibbe WE, Kingma T, Uyl-de Groot CA. Cost analysis and quality of life assessment comparing patients undergoing autologous peripheral blood stem cell transplantation or autologous bone marrow transplantation for refractory or relapsed non-Hodgkin’s lymphoma or Hodgkin’s disease. a prospective randomised trial. Eur J Cancer Oxf Engl 1990. 2001;37:1781–9.
Fagnoni P, Milpied N, Limat S, Deconinck E, Nerich V, Foussard C, et al. Cost effectiveness of high-dose chemotherapy with autologous stem cell support as initial treatment of aggressive non-Hodgkin’s lymphoma. PharmacoEconomics 2009;27:55–68.
Kouroukis CT, O’Brien BJ, Benger A, Marcellus D, Foley R, Garner J, et al. Cost-effectiveness of a transplantation strategy compared to melphalan and prednisone in younger patients with multiple myeloma. Leuk Lymphoma. 2003;44:29–37.
Jaime-Pérez JC, Heredia-Salazar AC, Cantú-Rodríguez OG, Gutiérrez-Aguirre H, Villarreal-Villarreal CD, Mancías-Guerra C, et al. Cost structure and clinical outcome of a stem cell transplantation program in a developing country: the experience in northeast Mexico. Oncologist. 2015;20:386–92.
Jones JA, Qazilbash MH, Shih Y-CT, Cantor SB, Cooksley CD, Elting LS. In-hospital complications of autologous hematopoietic stem cell transplantation for lymphoid malignancies: clinical and economic outcomes from the Nationwide Inpatient Sample. Cancer 2008;112:1096–105.
Gale RP, Ruiz-Argüelles GJ. The big freeze may be over: a contracting universe for cryopreservation? Bone Marrow Transpl. 2018;53:947–8.
Ruiz-Argüelles GJ, Gale RP. Autotransplantations Without Cryopreservation. J Glob Oncol. 2018;4 Accessed 6 Apr 2021. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6223471/.
Author information
Authors and Affiliations
Contributions
JMP was responsible for direct patient care and also data extraction and data analysis, interpreting the final results and writing the manuscript. ELR was also reponsible for direct patient care, interpreting the final results and manuscript review. ADA conducted data analysis, interpreted final results, wrote the manuscript, updated the reference list and created ‘summary of finding’ tables, writing the manuscript and manuscript review. CM, was responsible for designing the protocol and protocol supervision. CZO provided feedback on data extraction, data analysis, review of the statistical analysis and review of the manuscript. RRC conducted data extraction and submitted the research protocol for board ethic’s review. ACFM and AEL designed the protocol and conducted the apheresis procedures and stem cell harvest. JUAF, BMG, FRK and PS designed the protocol, were responsible for patient care, protocol supervision, interpreted the final results, helped writing the manuscript and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pessoa, J.M., da Rosa, E.L., Américo, A.D. et al. Cryopreserved versus non-cryopreserved stem cell autografts in multiple myeloma a restrospective cohort study. Bone Marrow Transplant 57, 1313–1318 (2022). https://doi.org/10.1038/s41409-022-01718-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-022-01718-2